Group 16
Reactions of non-metals with oxygen
Chapter overview
1 week
In this chapter learners encounter the reactions of the non-metals, carbon and sulfur [Note: the internationally accepted spelling is now 'sulfur', not 'sulphur',] with oxygen. Once again, the translation between word equation, picture equation and chemical equation is reinforced with exercises and examples.
There are no demonstrations prescribed by CAPS for this chapter, but we have included colourful photographs to provide learners with some experience of the splendour of these reactions. Since chemical reactions can seem quite abstract to learners and learning about them in isolation may seem to them as if it lacks relevance, we have also attempted to provide some real-world context to the examples in this chapter.
At the end of the chapter a few additional non-metals and their reactions with oxygen have been included as enrichment. You may choose to omit these, but they do highlight an important point, namely that not all non-metals form non-metal dioxides when they react with oxygen. Since both central examples of this chapter result in non-metal dioxides (viz. CO2 and SO2), one should be mindful of the possible introduction of a misconception in this regard.
4.1 The general reaction of non-metals with oxygen (0.5 hours)
4.2 The reaction of carbon with oxygen (1 hour)
|
Tasks |
Skills |
Recommendation |
|
Activity: Coal burning in air |
Drawing, balancing chemical equations |
Optional (Suggested) |
4.3 The reaction of sulfur with oxygen (1 hour)
|
Tasks |
Skills |
Recommendation |
|
Activity: The reaction between sulfur and oxygen |
Analysing, balancing chemical equations, drawing, modeling |
CAPS suggested |
4.4 Other non-metal oxides (0.5 hours)
This is an optional, extension section.
- What happens when a non-metal and oxygen react?
- What is the product called?
- How should we write equations for the reactions of carbon and sulfur with oxygen?
- Do all non-metals form dioxides with oxygen?
Oxygen is all around us in the air we breathe. It is a very reactive element. When an element is reactive, it means that it will readily react with many other substances. We saw evidence of the reactive nature of oxygen when we observed how it reacted with iron and magnesium to form metal oxides.
Substances that are not reactive are called unreactive or inert.
When a substance reacts with oxygen, we say it oxidises. It is called an oxidation reaction. Early chemists used the term oxidation for all reactions in which oxygen was a reactant. The definition of an oxidation reaction is broader and includes other reactions which you will learn about in Gr. 10 if you continue with Physical Sciences.
In this chapter we look at a few reactions of non-metals with oxygen. Where do we find non-metals on the Periodic Table?
On the right.
The general reaction of non-metals with oxygen
- reactive
- inert
- non-metal oxide
- oxidise
When a non-metal burns in oxygen, a non-metal oxide forms as product. Here is the word equation for the general reaction:
non-metal + oxygen → non-metal oxide
Can you see that it looks similar to the word equation for the reaction between a metal and oxygen? The only difference is that the word 'metal' has been replaced with 'non-metal' on both sides of the equation. Non-metal oxides have different chemical properties to metal oxides. We will learn more about this later on in the term.
An important chemical difference between metal and non-metal oxides is that when metal oxides dissolve in water, they form basic solutions and when non-metal oxides dissolve in water, they form acidic solution.
Let's look at a few specific examples of reactions in which non-metals react with oxygen. The first one is one that you are already familiar with, namely the reaction of carbon and oxygen.
The reaction of carbon with oxygen
- fuel
- fossil fuel
- generate
- non-renewable
- renewable
- alternative
Have you ever seen coal burning in air?

Coal is a form of carbon that is used as fuel for many different purposes. It is one of the primary fossil fuels that humans use to generate electricity for powering our industries, our activities and our living spaces. We will look at this in more detail next term in Energy and Change.
37% of the electricity generated worldwide is produced from coal.

Learners should have encountered the topics coal, fossil fuels, renewable and non-renewable energy sources and electricity generation in previous grades (specifically in Gr 6 and 7 Energy and Change) and they will also look at it again next term.
Coal burning in air
The energy in coal comes from the energy stored in plants and other organisms that lived hundreds of millions of years ago. Over the millennia, layers of dead plants and other biological waste were covered by layers of water and soil. The heat and pressure from the top layers caused the plant remains to turn into energy-rich coal.
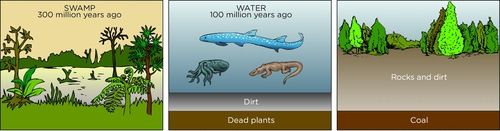
The energy released by burning coal is used to generate electrical energy in coal-powered power stations.
Coal is a form of carbon and when it burns in oxygen we can represent the reaction with the following word equation:
carbon + oxygen → carbon dioxide
Draw a picture equation for this reaction in the space below.
In the picture equation here, the black atoms are carbon (C) and the red atoms are oxygen (O). If learners do not have coloured pens or pencils, they can use patterns and shading to differentiate between different atoms.
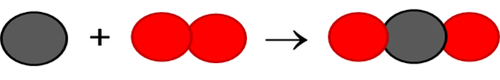
Convert the picture equation into a chemical equation. Is it balanced?
The chemical equation is C + O2 → CO2. This equation is already balanced.
What group is carbon in on the Periodic Table?
Group 14.
The other elements in the same group as carbon will react in the same way as carbon with oxygen.
Since coal is a non-renewable energy source as well as one that causes pollution and other environmental problems, scientists and engineers continue to look for alternative fuels and energy sources to eventually replace coal as an energy source. Can you think of the names of some alternative, renewable energy sources?
Wind energy, solar energy, biofuels, nuclear energy (the latter is not strictly renewable). This content is not included in CAPS (as part of the Grade 9 Matter and Materials requirements). The question has been included in an attempt to create some relevance. The topic of chemical reactions is an abstract one and it often helps to provide some real-world applications to link it to other, less abstract including learners' own experience. This will also link to what learners will cover next term in Energy and Change.
In the next section, we are going to look at the formation of another, less well-known, non-metal oxide named sulfur dioxide. Which non-metal do you think reacts with oxygen to form sulfur dioxide? See if you can write down the formula for sulfur dioxide before we carry on. Here is a hint: What does the di- in dioxide mean?
The non-metal is Sulfur (S). To write the formula, you may need to guide learners to realise there are 2 oxygen atoms in one molecule of sulfur dioxide: SO2
The reaction of sulfur with oxygen
- toxic
- preservative
What is the name of the product of the reaction between sulfur and oxygen? Use the name of the product and the general equation given at the start of the chapter to complete the following word equation:
sulfur + oxygen →
The equation should read: sulfur + oxygen → sulfur dioxide
A video on sulfur burning in oxygen
Sulfur burns in oxygen to form sulfur dioxide. Your teacher will not demonstrate this reaction, because the sulfur dioxide that forms is a poisonous gas that you and your classmates should not be exposed to.


A video () and a photo essay ( http://www.boston.com/bigpicture/2010/12/kawah_ijen_by_night.html) on sulfur mining.
Sulfur dioxide is sometimes used as a preservative for dried fruits, such as dried peaches and apricots and the guava rolls that so many of us love to eat. The fact that it is toxic means that very small quantities of it can be added to food to preserve it. In very small quantities SO2 does not permanently harm a large organism such as a human being, but bacteria cannot survive when it is present. Sulfur dioxide is also an important preservative in many South African wines.
The archaic (very old) name for sulfur is 'brimstone'.


In the following activity we are going to review word equations, picture equations and chemical equations, using the reaction between sulfur and oxygen as our context.
The reaction between sulfur and oxygen
You wrote the word equation for the reaction between sulfur and oxygen above. Did you write the following?
sulfur + oxygen → sulfur dioxide
QUESTIONS:
Sulfur (S) and oxygen (O2)
Sulfur dioxide (SO2)
S + O2 → SO2
When both sides of the reaction have the same number of the same types of atoms.
The reaction is balanced because it has the same number of S and O atoms on either side of the equation.
The picture equation for the reaction is shown below. Colours are not important as long as all atoms of the same element have the same colour. In our example, the sulfur atoms are yellow and the oxygen atoms are red. Chemists have evidence that the actual shape of the SO2 molecule is not linear; in other words, the atoms do not all lie in a straight line as they do in CO2. Rather, the SO2 molecule is believed to have a bent shape, as it appears in the picture equation. Since the explanation for this shape requires some understanding of bonding models, which are beyond the scope of this level of the curriculum, we recommend that you do not 'go there', but accept 'linear' drawings of the SO2 molecule as correct.

- Use play dough or clay to build models of the reactants and products of the reaction. This is what your starting reactants could look like:

Provide play dough or modelling clay for this part of the activity. You can also get learners to construct the equation by using sheets of white paper and drawing a '+' and '\(\rightarrow{}\)

And then they must rearrange the atoms to form the product as shown here:

You can also repeat this exercise with the other reactions covered so far.
Challenge question: How many bonds were broken and how many bonds were formed during this reaction?
1 bond was broken (between the oxygen atoms) and 2 bonds were formed (1 between each of the oxygen atoms and the sulfur atom)
This is an extension question.
Other non-metal oxides
Important note: This is not required by CAPS but is offered as enrichment.
- dioxide
- systematic name
You might not cover this section in class as it is an extension.
We have looked at two examples of non-metals reacting with oxygen to form non-metal oxides. Both of our examples had a dioxide as product (carbon dioxide and sulfur dioxide). Do all non-metals form non-metal dioxides when they react with oxygen? What do you think?
Get learners to discuss this for a moment. You may want to ask if water (H2O) is a non-metal oxide. Point out that hydrogen (H) is a non-metal and that water should be a non-metal oxide. Is it a dioxide? No, because it contains only one oxygen. This is important in highlighting how the non-metals in different groups react differently with oxygen.
Not all non-metal oxides are dioxides, as the following examples show.
The reaction between phosphorus and oxygen
Phosphorus is a very reactive non-metal. Can you remember what reactive means?
Ask your learners this question to see what they can remember. Compounds that are reactive will readily react with many other substances.
Video on phosphorus burning in oxygen
When phosphorus reacts with oxygen the chemical equation for the reaction is the following:
4 P + 5 O2>→ 2 P2O5
How many phosphorus atoms are in P2O5? How many oxygen atoms are in P2O5?
There are 2 phosphorus and 5 oxygen atoms.
What is the systematic name of the product of this reaction? (If you are unsure how to name it, sneak a peek at the first chapter!)
Diphosphorus pentoxide
Can you write a word equation for this reaction?
phosphorus + oxygen → diphosphorus pentoxide
Our final example is a compound that you should be very familiar with!
The reaction between hydrogen and oxygen
Hydrogen and oxygen also react spectacularly. The reaction between a large quantity of hydrogen and oxygen in the air produces a beautiful orange fireball and a very loud boom! (You can watch the video in the visit box to see this in slow motion.)
A slow motion video of a hydrogen-filled balloon exploding in air
Here is a diagram to show what is really happening to the compounds in this reaction. The purpose of the candle shown in the picture is to set the hydrogen gas alight, in other words: to provide enough energy for the reaction to start.
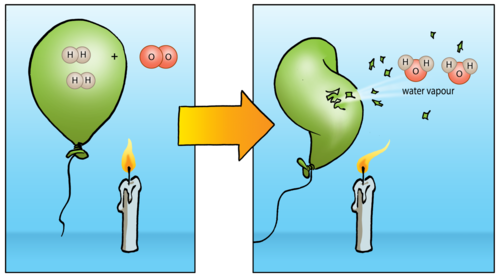
If you choose to demonstrate this in class, take note of the safety precautions. This is a very explosive reaction and everyone present should wear safety goggles.
Can you complete the following chemical equation? The reaction is between hydrogen and oxygen. Write the product where it belongs.
2 H2+ O2→
2 H2 + O2 → 2 H2O. You can also get learners to practice rearranging the atoms with this equation, making sure they have a balanced equation.
What is the common name of the product of this reaction? What is the systematic name of the product of this reaction? (If you are unsure how to name it, sneak a peek at the first chapter!)
The common name is water and the systematic name is dihydrogen monoxide.

The reaction of hydrogen with oxygen (video)
In this chapter we learnt about some of the reactions between non-metals and oxygen. Some of the skills that we practised during this chapter were: writing equations (word, picture and chemical equations) and naming compounds.
Summary
- Non-metals react with oxygen to form non-metal oxides.
-
The non-metal and oxygen gas (O2) are the reactants in this type of reaction, and a non-metal oxide is the product.
- The reactions of carbon and sulfur with oxygen are examples of non-metals reacting with oxygen.
- Carbon and sulfur both form dioxides with oxygen, but this is not true of all non-metals.
Concept map
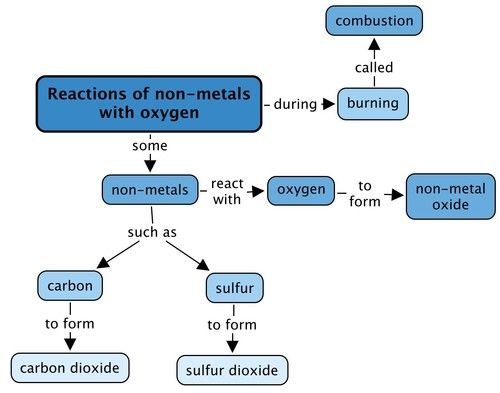
Complete the concept map below. What will you fill in for the products when the two different non-metals react with oxygen during combustion?
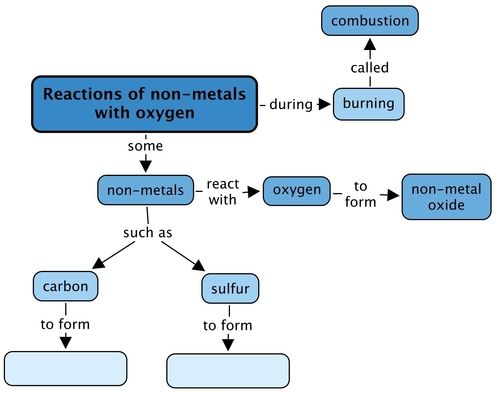
This is the completed concept map.
Revision questions
Fill in the missing words in these sentences. Write the word on the line below. [5 marks]
-
A substance that will react readily with many other substances is called a ____________ substance.
-
Substances that do not react with other substances and do not change into other compounds are called ____________ or ____________.
-
When a non-metal reacts with oxygen the product of the reaction is a ____________.
-
When a compound reacts with oxygen, we say it has become ____________.
- reactive
- unreactive or inert
- non-metal oxide
- oxidised
-
Write a short paragraph (3 or more sentences) to explain what you understand each of the following terms to mean, in your own words. [3 x 3 = 9 marks]
systematic name
Learner's paragraph should contain at least the following ideas:
- The systematic name of a compound is the name that is recognised by IUPAC.
- IUPAC refers to the International Union of Pure and Applied Chemistry.
- The systematic name of any given compound should be unique so that the compound cannot be confused with any other compound.
preservative
Learner's paragraph should contain at least the following ideas:
- A preservative is a chemical compound that is added to a product (such as a foodstuff or a beverage) to make it last longer.
- Most preservatives are poisonous to microorganisms, but are added in such small quantities that they are not harmful to humans.
-
SO2 is used as a preservative in many foods, including dried fruit and wine.
non-renewable energy source
Learner's paragraph should contain at least the following ideas:
- Non-renewable energy sources refer to sources that can be used up, such as fossil fuels.
- Coal, oil and natural gas are examples of non-renewable fuels.
- The energy in these energy sources comes from the energy stored in plants and other living organisms that were fossilised over millions of years.
-
For each of the following reactions, complete the tables by providing the missing equations.
The reaction between carbon and oxygen [6 marks]
Word equation
Chemical equation
Picture equation
Word equation
carbon + oxygen → carbon dioxide
Chemical equation
C + O2 → CO2
Picture equation

The reaction between sulfur and oxygen [6 marks]
Word equation
Chemical equation
Picture equation
Word equation
sulfur + oxygen → sulfur dioxide
Chemical equation
S + O2 → SO2
Picture equation

Total [26 marks]
