The atmosphere
Chapter overview
2 weeks
The first chapter in the Planet Earth and Beyond strand looked at the spheres of the Earth. The atmosphere was briefly introduced in this chapter and is now covered in more detail.
The atmosphere is the layer of gases around the Earth. The important concepts that need to be understood in this chapter are:
- the air we breathe does not contain 'nothing', but rather an important mix of gases which support and sustain life on Earth
- gravity causes the density gradient in the atmosphere, and makes the atmosphere 'stick' to Earth
- the temperature in the atmosphere varies with altitude as a result of radiation from the Earth, the specific gases present at different altitudes, chemical reactions taking place in the atmosphere, and the energy from the Sun.
The concept of space and the 'nothingness' that we breathe might be difficult for learners to comprehend. We have included a number of models and pictures to help them visualise the vastness of space, and understand that the atmosphere, which surrounds us, is required for life on this planet.
A common misconception is that land plants generate most of the oxygen in the atmosphere, whereas this actually occurs in the oceans where \(\text{70}\)% of the planet's oxygen supply is produced.
Direct sunlight does not heat the atmosphere. Only a small portion of the heating can be accounted for by direct sunlight, most of the heating is through conduction, convection and re-radiation of the Sun's energy.
Another misconception is that greenhouse gases make up a major part of the atmosphere. Nitrogen and oxygen make up \(\text{99}\)% of the atmosphere while other gases, including greenhouse gases, make up only about \(\text{1}\)%.
4.1 What is the atmosphere? (1 hour)
|
Tasks |
Skills |
Recommendation |
|
Activity: How thick is the atmosphere compared to the size of the Earth? |
Measurement, interpretation, representing information |
Suggested |
4.2 The troposphere (0.5 hours)
|
Tasks |
Skills |
Recommendation |
|
Activity: Drawing a graph of the temperature gradient in the troposphere |
Drawing graphs, doing calculations, measuring |
CAPS suggested |
4.3 The stratosphere (0.5 hours)
4.4 The mesosphere (0.5 hours)
4.5 The thermosphere (1 hour)
|
Tasks |
Skills |
Recommendation |
|
Activity (model building): How thick are the layers of the atmosphere? |
Building a model, drawing diagrams, data handling |
Suggested |
4.6 The greenhouse effect (2.5 hours)
|
Tasks |
Skills |
Recommendation |
|
Activity: Comparing the Earth, Mars and Venus |
Interpreting data, providing explanations, application |
Suggested |
|
Investigation: A model of the greenhouse effect |
Building a model, data collection, data analysis, interpreting results, drawing conclusions |
CAPS suggested |
|
Investigation: Ice core analysis |
Interpreting data, formulating, investigative questions, drawing conclusions, representing data |
Suggested |
- What is the atmosphere?
- What makes up the atmosphere?
- Does the atmosphere change as you go further from the Earth's surface?
- Can the atmosphere be divided into different layers?
- Where does the atmosphere end?
- What important aspect of the atmosphere allows life to exist on earth?
- What is the greenhouse effect?
- How do humans contribute to the greenhouse effect?
In the first chapter of Planet Earth and Beyond, you learnt about the different spheres of the Earth. The atmosphere was mentioned briefly in Chapter 1. In this chapter we will look at the atmosphere in more detail.

What is the atmosphere?
- atmosphere
- troposphere
- stratosphere
- mesosphere
- thermosphere
- exosphere
- altitude
- temperature gradient
Reveal Earth's atmosphere.
The atmosphere is the layer of gases which surrounds the Earth. It contains the following mixture:
- nitrogen (78,08%)
- oxygen (20,95%)
- argon (0,93%)
- carbon dioxide and other trace gases (0,04%)
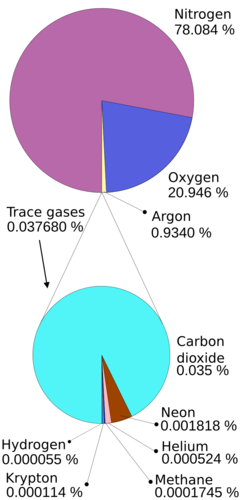
The gas molecules in the atmosphere are kept close to the Earth by gravity. The effect of gravity means that there will be more gas molecules closer to the Earth's surface than further away. As you move further and further away from the surface of the Earth, the gas molecules become fewer and the spaces between the molecules become larger, until there are no more gas molecules and only spaces left. The atmosphere therefore does not have a set boundary, but rather fades away into space.
Sports teams and athletes need to acclimatize when they get to a new location at altitude, before performing, so that their bodies can get used to the lower level of oxygen.
Some endurance athletes spend several weeks training at high altitudes, preferably 2400 m above sea level, so that their bodies adapt by producing more red blood cells. This gives them a competitive advantage when returning to a lower altitude to compete.
When we walk up a very high mountain, there is less oxygen present. We may feel out of breath. People sometimes say that the air is thinner higher up. When they say this they mean that there is a lower concentration of oxygen molecules.
The density of the atmosphere decreases with an increase in the height above sea level (altitude). Density is an indication of how many particles are present in a specific volume of gas. When the density is high, there are lots of gas molecules present. If the density is low, there are fewer gas molecules present.
The atmosphere is a very important part of the Earth. It keeps the planet warm and protects us from the harmful radiation of the Sun. It also ensures a healthy balance between oxygen and carbon dioxide so that life can be sustained on the planet.
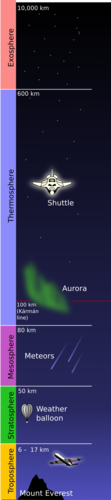
The atmosphere has four main layers. We start measuring these from sea level and move towards space. The diagram alongside illustrates this. The surface of the Earth is at the bottom of the diagram, with Mount Everest drawn in. The first layer is the troposphere, then the stratosphere, the mesosphere and the thermosphere. Above the thermosphere, the atmosphere merges with outer space in the layer known as the exosphere.
The exosphere is not considered part of the atmosphere due to the very low density of gases, but it is still briefly discussed in this chapter. In some other resources which you may look at, the exosphere is sometimes discussed as a layer or upper limit of the atmosphere.

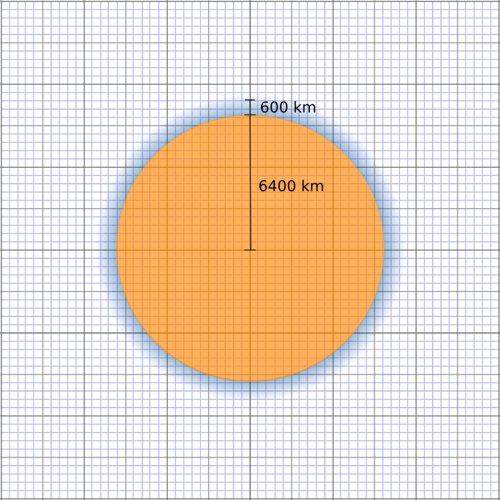
The atmosphere is actually a very thin layer compared to the size of the Earth. It is almost like the skin of an orange, relative to the size of the orange.
A journey through the atmosphere.
How thick is the atmosphere compared to the size of the Earth?
INSTRUCTIONS:
Answer the following questions to help you draw the diagram.
-
What is the radius of the Earth? Choose an appropriate scale and draw a circle in your notebook to represent the Earth.
-
How thick is the atmosphere in km? Use the same scale as above and draw the atmosphere around the Earth.
-
Indicate the atmosphere density gradient on your diagram.

-
6400 km.
-
About 480-600 km (accept any answer within this range).
-
Learner-dependent answer.
The atmosphere is denser closer to the surface than further away. This can be indicated by using darker going to lighter colours, or using more dots and fewer dots, or any other way of indicating that the atmosphere is denser at the bottom. A scale needs to be included. A possible diagram could look like this:
Scale drawing of the Earth and the surrounding atmosphere. 1 block = 400 km.
Scale drawing showing the thickness of the atmosphere relative to the thickness of the Earth.
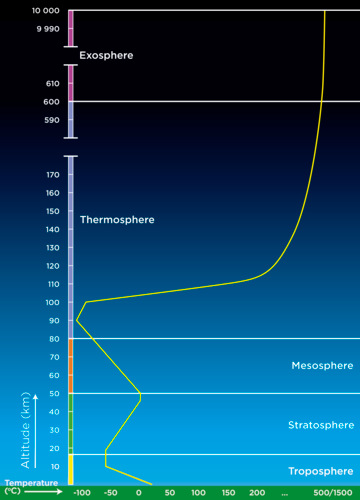
Each layer of the atmosphere has a different temperature gradient, in other words, the temperature changes gradually as you move through each layer. The following graph shows how the temperature changes as you move through the atmosphere. The layers of the atmosphere are also indicated on the graph. Temperature is on the x-axis and altitude is on the y-axis. The red line shows the change in temperature. Note that as you move further to the left on the graph it is colder, dropping far below 0 °C, and further to the right is hotter, reaching over 1000 °C.
Altitude is a measure of height above sea level.
Our atmosphere is escaping.
Now let's look at each of the layers of the atmosphere.
The troposphere
The boundaries between the layers in the atmosphere are not as clear as the boundaries between liquids. The values are often given as a range and the transitions take place in zones called pauses, for example the tropopause is the zone in-between the troposphere and the stratosphere.
The troposphere is the lowest layer in the atmosphere. It stretches from sea level up to about 9 km at the poles and 17 km at the equator. Due to the Earth's rotation, the atmosphere is thicker at the equator than at the poles. On average it is about 12 km thick.
The density of air decreases as you move further away from the surface of the Earth. The first two layers of the atmosphere contain most of the mass of the atmosphere. The bottom part of the troposphere has a high enough density for us to breathe and is the layer of the atmosphere in which we live.

Troposphere comes from the Greek word tropein, meaning to change, circulate or mix.
The air in the troposphere is in constant motion. As it is warmed by the Earth, the warm air moves away and gets replaced by cooler air which travels in convection currents. This is the basis for cloud formation and weather patterns. All the Earth's weather systems take place close to the Earth in the troposphere.


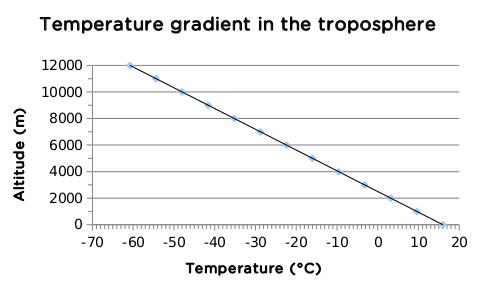
The temperature in the troposphere decreases with altitude - the further you move away from the surface, the colder it becomes. The temperature decreases about 6,4°C for every kilometre increase in altitude. In the following activity you will investigate the change in temperature as height above sea level increases.
Drawing a graph of the temperature gradient in the troposphere
If you would like to assess learners' tables and graphs you can use Assessment Rubric 3 and 4 to do so. As an extension you could get the learners to draw a similar graph for the change in pressure as you move through the troposphere. They will have to first find out what the pressure gradient is in the atmosphere.
INSTRUCTIONS:
- Using the information in the previous text, set up your own table displaying the temperature change in the troposphere from 0 - 12 km.
- Then draw a neat, accurate graph of this data.
-
Assume that the average temperature on the surface of the Earth is 16°C.
- Choose an appropriate scale for the x- and y-axes of your graph.
- Label the axes and give the graph a heading.
Use the following space to draw a table for your data.
An example table is given below:
Change in temperature as altitude increases in the troposphere
|
Altitude (m) |
Temperature ( o C) |
|
0 |
16 |
|
1000 |
9.6 |
|
2000 |
3.2 |
|
3000 |
-3.2 |
|
4000 |
-9.6 |
|
5000 |
-16 |
|
6000 |
-22.4 |
|
7000 |
-28.8 |
|
8000 |
-35.2 |
|
9000 |
-41.6 |
|
10 000 |
-48 |
|
11 000 |
-54.4 |
|
12 000 |
-60.8 |
Use the following space to draw your graph.
An example graph is given below.
An alternative graph with the altitude on the x-axis is shown below:
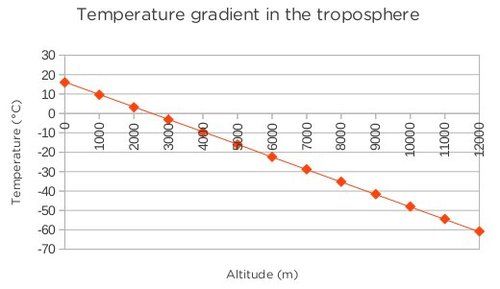
The temperature in the troposphere decreases steadily until it reaches about -60°C at about 10-12 km above sea level. The temperature here stabilises before it increases again. This is the transition zone between the troposphere and the stratosphere. This layer forms an invisible barrier which prevents the warmer moist air from escaping from the troposphere. Beyond this region air does not circulate much and weather patterns are not found any more.
Layers of the atmosphere.
The stratosphere
- ozone
- CFCs
The stratosphere is the layer above the troposphere. It stretches from 12 km to 50 km above the surface of the Earth. 90% of the mass of the atmosphere is found in the troposphere and the stratosphere.
Aeroplanes fly in the lower stratosphere because the air is much more stable than in the troposphere. The density of the air in the stratosphere is very low and decreases with altitude.
Scientists use weather balloons to gather information on the temperature and pressure as they move up from the Earth's surface to the stratosphere. A weather balloons carries a small device, called a radiosonde, which sends back information on atmospheric pressure, temperature, humidity and wind speed.
Weather balloons were first used 70 years ago, and are still the key instrument for meteorologists to assess and predict the weather. This information is used in many ways, for example, to compile the weather report on TV or to warn of flooding or issue a hurricane warning.


Footage from a weather balloon.
Weather balloons are filled with helium or hydrogen and rise higher and higher into the atmosphere. Do you think they continue rising up for ever? What do you think happens to the balloon as it increases in altitude? Hint: Think of what happens to the gas inside the balloon as the altitude increases. Discuss this with your class and take some notes below.
Discuss this with your class as it provides a good opportunity to revise some of the concepts learned in Gr 8 on the particle model of matter, as well as extend this knowledge. As the weather balloon rises higher and higher in the atmosphere, the pressure decreases. The volume of gas inside the balloon therefore increases and the balloon expands. Eventually it gets to a point where the balloon material cannot stretch anymore and the balloon bursts. This usually happens between 25 and 30 km above the Earth's surface. You can also explain to learners that the radiosonde has a small parachute attached to it which opens up when it begins to fall back to Earth. This is to slow its fall so that it does not crash down and cause damage to someone or something. Also, these sensors are never retrieved. Scientists don't need to retrieve them to get the information back because data will have been sent by radio signals.
NASA is thinking of sending high altitude weather balloons to probe the atmosphere of Mars.
Ozone gas (O3) is found in the stratosphere. Ozone gas is made up of ozone molecules. Each molecule consists of three oxygen atoms. Ozone plays an important role in absorbing harmful UV rays from the Sun by forming, breaking down and reforming ozone molecules over and over again. When UV light reaches the Earth, it can cause cancer, affect plant growth, and the life cycles of species.
You should always take necessary precautions against the damaging effects of UV light to your skin and eyes. Wear sun cream and a hat when outside and avoid spending hours in the sun without protection.
What happens to ozone in the atmosphere?
This is extension content on what happens to ozone in the atmosphere and is not prescribed by CAPS. However, it is within the learners' capacity to understand it, given what they have learned in Matter and Materials. For more information about ozone you can visit http://www.ozonedepletion.info/education/ozone.html
The formation and destruction of ozone is a natural process that takes place in the stratosphere. Oxygen forms ozone, and ozone breaks apart again to form oxygen. The following diagram shows the reactions that take place.
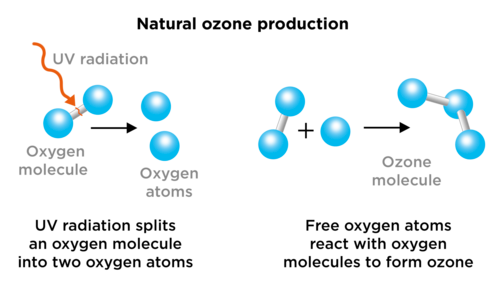
What holds the oxygen atoms together in a molecule?
A chemical bond.
What is the term given to a molecule of oxygen which consists of two atoms of the same element bonded together?
A diatomic molecule.
Although ozone is considered a pollutant in the troposphere, in higher altitudes in the stratosphere, ozone is considered vital as it protects Earth from too much ultraviolet radiation.
The ozone reactions lead to the heating of the stratosphere, increasing the temperature from -60°C to about 0°C. As a result, the air becomes warmer as you move further away from the Earth in the stratosphere.
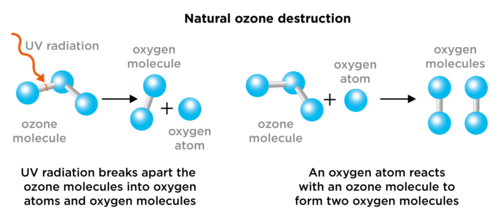
The problem comes in when there are molecules present which interfere with these natural processes. Chlorofluorocarbons, or CFCs, are molecules which release chlorine atoms into the stratosphere. Chlorine atoms react with ozone, destroying it before it can absorb harmful UV rays. The following diagram show how CFCs react with ozone.
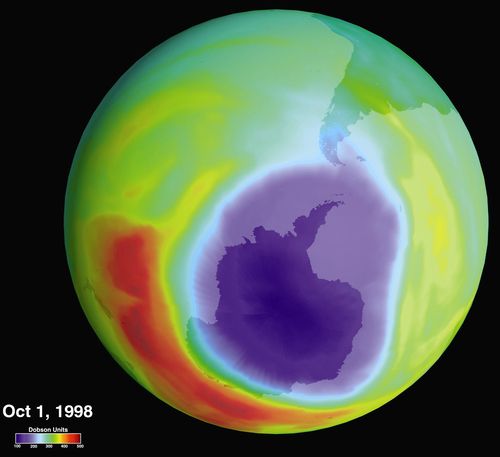
Theozone hole is an annual thinning of the ozone layer over Antarctica, caused by chlorine from CFCs in the stratosphere.
Ozone layer danger.
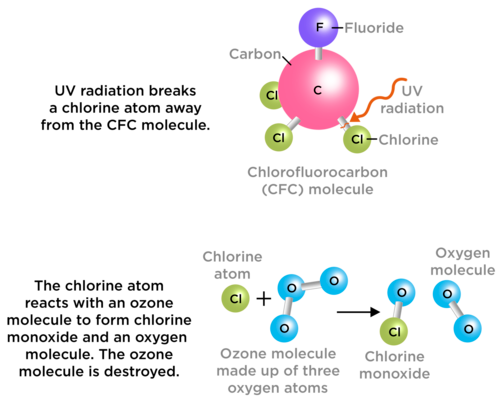
CFCs used to be found in aerosols and refrigerator gas, and were given off by industrial processes. Scientists noticed that these gases interfered with ozone. This could have had a serious impact on life on Earth and the use of CFCs was banned.
On 14 October 2012 Felix Baumgartner set a world record by jumping from an altitude of about 39 km - from the stratosphere to the Earth. He is the first man to jump from the stratosphere. It took him nine minutes to reach Earth and he reached a maximum speed of 1343 km/h.
Watch a video of Felix's jump here.
The mesosphere
- meteor
- meteorite
The mesosphere extends from around 50 km to 80 km above the Earth's surface. The atmosphere reaches its lowest temperature (-90 °C) in the mesosphere. The air density is extremely low, but there is still enough air to burn up rocks and dust entering from space.
Watch the meteor shower over Johannesburg four years ago and short clips of the meteor show that hit Russia in early 2013
A meteor is a rock that enters the atmosphere from space. It travels at extremely high speed, up to 30 000 m/s. As a meteor enters the atmosphere, the air in front of it is compressed. The air heats up and the meteor burns up as a result of heat and friction. When we look up at the night sky, we might see a streak of light flashing for a brief moment. This is commonly called a shooting star, but is in fact a meteor burning up in the mesosphere.

Most meteors are fairly small and burn up completely while whizzing through the mesosphere. Some of the larger, denser meteors can reach the Earth and are then called meteorites. When the meteorite strikes the ground, it kicks up dust and soil and leaves an impact crater on the Earth's surface. The size of the crater depends on the size, density and speed of the meteorite.
The biggest meteorite impact crater on Earth is found at Vredefort in South Africa. The crater is 300 km wide and was formed 2023 million years ago, long before dinosaurs lived on the planet.


The thermosphere
- ionosphere
- aurora
- northern lights (aurora borealis)
- southern lights (aurora australis)
- International Space Station
The thermosphere is the layer of the atmosphere from 80 km upwards. The density of the air is extremely low. The further away you move from the Earth, the less dense the concentration of molecules becomes until the atmosphere becomes space.
Most satellites that we depend on every day are in Low Earth Orbit (LEO), orbiting the Earth at an altitude between 160 km and 2,000 km. The International Space Station (ISS) is situated at 370 km in the thermosphere. This is an international facility in space that is used for research purposes.
In 2002, Mark Shuttleworth became first South African in space when he launched with a Russian space mission. He spent eight days on board the the International Space Station, participating in experiments related to AIDS and genome research.

You can see the ISS in the sky if you know where to look. The following link will give you the locations. http://spotthestation.nasa.gov/sightings/index.cfm?country=South_Africa®ion=South_Africa_None#.UhyQGZIwdXF
The temperature in the thermosphere increases from - 90°C to as high as 1500°C. The thermosphere is very sensitive to an increase in energy and a small change in energy results in a high temperature increase. At times of increased solar activity, the temperature can easily increase up to 1500°C. However, the thermosphere will feel cold as there are few particles present to collide with our skin and transfer enough energy for us to feel the heat.
High energy light (for example, UV light) can cause atoms or molecules to lose electrons, forming ions. The region where this takes place is called the ionosphere. The ionosphere is found mainly in the thermosphere. The Sun also gives off charged particles (the solar wind), which can enter the Earth's atmosphere (mostly near the poles) and react with the ions and electrons in the ionosphere, causing a phenomenon called the aurora. It is a colourful display of light in the sky at the poles. In the northern hemisphere, it is called the northern lights or aurora borealis, and the southern lights or aurora australis in the southern hemisphere.
The ionosphere reflects longer wavelength radio waves, for example the radio waves we use for radio and surface-broadcast television (not satellite television), allowing the signal to be broadcast over a larger distance. The ions in the ionosphere also absorb ultraviolet radiation and X-rays.
The region beyond the thermosphere is called the exosphere. This layer has very few molecules and extends into space.

The structure of our atmosphere.
How thick are the layers of the atmosphere?
In this activity you will build a model to represent the different layers of the atmosphere. In addition to the model, you need to draw an accurate diagram in your workbook to represent the thickness of each layer. Use a ruler to draw an accurate scale diagram.
This activity demonstrates the relative thickness of the layers of the atmosphere. Learners might not realise how thin the troposphere, the layer in which we live, is compared to the rest of the atmosphere. After this activity they should have a better comprehension of the thickness of the layers of the atmosphere relative to each other.
MATERIALS:
- large measuring cylinder or tall drinking glass
- corn kernels (popcorn)
- samp
- dried peas
- beans

Alternative materials are different types of dry breakfast cereals, or different shapes of pasta (shells, screws, macaroni, gnocchi, etc. Any tall, thin transparent container can be used.)
INSTRUCTIONS:
- Add a 0,5 cm layer of dried split peas to represent the troposphere (1 layer of peas thick).
- Add a 1,5 cm layer of corn kernels on top of the peas to represent the stratosphere.
- Add a 1,5 cm layer of samp on top of the corn kernels to represent the mesosphere.
- Add a 24 cm layer of beans on top of the samp to represent the thermosphere.


You will notice that the area where the two layers meet is not always clear cut. The kernels might have mixed a little bit. The atmosphere is the same. There is a not a clear line separating two layers, but they mingle in the area of contact.
Table showing the heights of the layers in Earth's atmosphere and in the model
|
Layer |
Represented by |
Height of layer (km) |
Height of layer (cm) |
|
Troposphere |
Dried split peas |
~10 |
0,5 |
|
Stratosphere |
Corn kernels |
~30 |
1,5 |
|
Mesosphere |
Samp |
~30 |
1,5 |
|
Thermosphere |
Beans |
~480 |
24 |
QUESTIONS:
Draw a labelled diagram of the model using the graph paper. Include a scale. The density of the atmosphere decreases with altitude. Show this on your diagram as well.

Learners can either draw a diagram here on the graph paper in their workbooks or on another piece of larger paper. If they draw it here in their workbooks, they will not be able to draw the diagram using the same scale as the model, as there is not enough space to draw a diagram 27,5 cm long. They can do this on another separate piece of paper. Using the graph paper, they must indicate what scale they have used, for example, 1 block or cm represents 20 km, etc.
To indicate density, learners can use darker and lighter colours or use lots of dots at the bottom and gradually fewer dots higher up.
What atmospheric layers are represented by the different grains in the model?
This answer depends on the model made.
Peas - troposphere
Corn - stratosphere
Samp - mesosphere
Beans - thermosphere
In the model in the activity, how many kilometers does 1 cm represent?
This can be calculated, for example: 10 km / 0,5 cm = 20 km/cm.
How much thicker is the stratosphere compared to the troposphere?
It is three times thicker.
How much thicker is the thermosphere compared to all the other layers combined?
It is seven times thicker.
Where in this model would you expect to find clouds?
Troposphere
Where in this model would you expect to find the Drakensberg Mountains?
Troposphere
Where in this model would you expect to find a satellite?
Thermosphere
Where in this model would you expect to find meteors burning up?
Mesosphere
In which layer is there life? What is different about this layer?
Troposphere. It is the thinnest layer.
The greenhouse effect
- greenhouse gases
- greenhouse effect
- global warming
- climate change
- carbon dioxide
- methane
- water vapour
- radiation
You have learned a lot about greenhouse gases in Natural Sciences. In this section we will be looking at how important greenhouse gases are to sustain life on Earth.
Earth's atmosphere contains mostly (99%) nitrogen and oxygen, but a small percentage (1%) of the atmosphere contains gases like water vapour (H2O), carbon dioxide (CO2) and methane (CH4). Carbon dioxide is a product of respiration in all organisms and also a gas given off by industrial processes and the burning of fossil fuels and vegetation. Methane is a gas, also called natural gas, which occurs in reservoirs beneath the surface of the Earth. It is also given off by decomposing plant and animal material and animals give off methane as part of their digestion. Water vapour is formed when water evaporates on Earth.
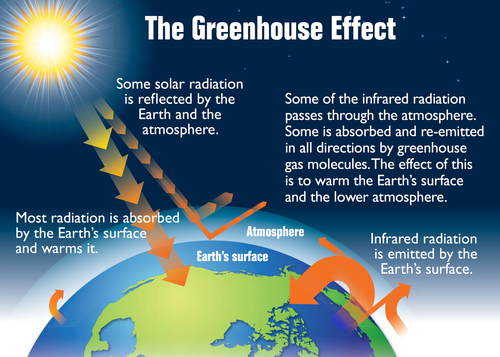
Water vapour, methane and carbon dioxide are gases which let through incoming visible light from the Sun. The incoming radiation from the Sun is absorbed by the Earth's surface and warms it. The Earth's surface emits infrared radiation. Infrared radiation is absorbed by the greenhouse gases and re-emitted in all directions. This increases the temperature of the Earth's surface and lower atmosphere, above what it would be without the gases, called the greenhouse effect. These gases are very important to regulate the Earth's temperature.
Learn more about the greenhouse effect with this simulation. http://phet.colorado.edu/en/simulation/greenhouse
As you can see in the diagram, the radiation from the Sun is able to reach the Earth and warm it up. The energy that is given off by the Earth is trapped by the water vapour, carbon dioxide and methane. This ensures that the Earth stays warm. It is almost as if the gases form a blanket around the Earth keeping some of the heat inside. The gases are referred to as greenhouse gases. A greenhouse is a glass structure that is used to grow plants. The glass lets the heat of the Sun through, but then keeps the heat inside the structure so that the plants have a moderate climate in which to grow. Water vapour, carbon dioxide and methane act in the same way.
Another analogy of the greenhouse effect is to think about a car parked in the Sun with the windows shut. After awhile it will heat up inside the car.
The Earth is a very unique planet due to the make-up of its atmosphere. In this chapter you have learned about the composition of the Earth's atmosphere. Let us compare the atmosphere of Earth to its neighbouring planets, Mars and Venus.
Comparing Earth, Mars and Venus

INSTRUCTIONS:
- The table below gives information about the gases in the atmospheres of the three planets: Venus, Earth and Mars.
- Study the table and answer the questions that follow.
Percentage of gases making up the atmospheres of Venus, Earth and Mars.
|
Venus |
Earth |
Mars |
|
|
Carbon dioxide (CO2) |
96,5% |
0,03% |
95% |
|
Nitrogen (N2) |
3,5% |
78% |
2.7% |
|
Oxygen (O2) |
Trace |
21% |
0,13% |
|
Argon (Ar) |
0,007% |
0,9% |
1,6% |
|
Methane (CH4) |
0 |
0,002% |
0 |
QUESTIONS:
Compare the data for Venus and Earth. What similarities and difference do you notice?
Venus' atmosphere is made up of mainly carbon dioxide. Earth's atmosphere contains very little carbon dioxide.
Earth's atmosphere is made up of mainly nitrogen and Venus has very little nitrogen.
Earth's atmosphere contains a lot of oxygen, whereas Venus contains only trace amounts (very little).
Argon is present at a low level for both planets.
Venus has no methane.
Compare the data for Venus and Mars. What similarities and difference do you notice?
Venus and Mars have very similar atmospheres. Most of their atmospheres are made up of carbon dioxide, with very low levels of all the other gases. Neither of them contain methane.
What is the biggest difference between Earth's atmosphere and the atmospheres of the other two planets?
Earth's atmosphere contains almost no carbon dioxide, but has a lot of nitrogen and oxygen, whereas the atmospheres of Mars and Venus consist primarily of carbon dioxide and a few other gases.
Why is the level of oxygen so much higher on Earth than on the other two planets?
Oxygen is produced by the oceans and plants on Earth. Venus and Mars have no liquid water and there is no life on the planet, so oxygen cannot be produced in large quantities.
Why do you think there is no methane gas on Venus and Mars?
Methane gas is produced by animals and by decomposing plant and animal matter. There is no life on Venus and Mars and nothing present on these planets which can produce methane.
Predict whether you think the temperature on the surface of Venus will be low or high. Give reasons for your answer.
The temperatures will be very high. Venus has a lot of carbon dioxide which traps the heat of the Sun and makes the surface temperature high.
Venus is closer to the Sun than the Earth is, and therefore receives more radiation.
Carbon dioxide and the greenhouse effect.
The atmospheres of Venus and Mars are very similar. Both planets have mainly carbon dioxide in the atmosphere, and very little other gases. However, the two planets are quite different.
Venus has a very dense atmosphere which results in a high concentration of carbon dioxide on its surface. This causes an extreme greenhouse effect and very high temperatures on the surface of Venus. Venus has an average surface temperature of 462 °C. This is too high to sustain life as we know it.
Venus is the hottest planet in the solar system; the temperature is hot enough to melt lead.
Mars, on the other hand, has almost no atmosphere, so, although there is carbon dioxide present, the density is very low and almost no greenhouse effect takes place. Mars is also much further away from the Sun. It is a very cold planet, with an average temperature of -55°C. This is too low to sustain life as we know it.
Earth has the right composition of atmospheric gases to sustain life. It has the right balance between oxygen and nitrogen so that plants and animals can breathe, and just enough carbon dioxide and methane to keep the planet warm enough so that life can be sustained. Many scientists think that it is the life on Earth that keeps the atmosphere in this perfect balance. Plants produce oxygen and re-circulate carbon dioxide on Earth. They believe that if life were to disappear from Earth, the atmosphere would become like Mars or Venus.
A model of the greenhouse effect
A greenhouse gas demonstration.
In the greenhouse effect, carbon dioxide traps the heat of the Sun. In this investigation, you will use bottles with air and carbon dioxide, respectively, to model the greenhouse effect. You are going to investigate the following question: Does air or carbon dioxide absorb more heat?
CAPS prescribes plastic bags, we have used plastic cold drink bottles as it makes data collection easier.
You can make the holes in the lids of the bottles beforehand using a knife or by hammering a nail in through the lid. Otherwise learners will need to do this and you will need a hammer, a nail about the width of the thermometer and a wooden block to hammer into.
You can prepare and collect small bottles of carbon dioxide beforehand, otherwise learners will need to do this. To collect a bottle of carbon dioxide, add one tablespoon of bicarbonate of soda to the small bottle. Add 10-20 ml of vinegar and place the lid back on. Hold the mouth of the small bottle over the CO2 container and pour the carbon dioxide into the large bottle. Carbon dioxide is denser than air and can therefore be poured into the large bottle. Add more vinegar when the effervescence stops. Repeat 2-3 times until the large bottle is full. If a burning match at the mouth of the bottle goes out immediately, the bottle is full.
AIM: Write an aim for this investigation.
A possible answer is 'To determine whether carbon dioxide or air traps more heat.
HYPOTHESIS: Write a hypothesis for this investigation.
Learner-dependent answer.
MATERIALS AND APPARATUS:
- two glass bottles or clear cold drink bottles with lids
- 2 thermometers
- Prestik
- heat source (two study lamps)
- vinegar
- bicarbonate of soda
- small cold drink bottle with lid
METHOD:
Set up the experiment as in the photograph.

-
Mark one bottle as 'Air' and the other bottle as 'CO2'.
- If the lids do not have the thermometers in them already, prepared by your teacher, make a hole in each of the lids. You can do this using a hammer and nail and hammering the nail through the lid into a wooden block. Secure the thermometer in each lid. You can use Prestik to do this.
- Fill the first bottle with air and secure the thermometer and close the lid tightly.
- Fill the second bottle with carbon dioxide:
- To collect a bottle of carbon dioxide, add one tablespoon of bicarbonate of soda to the small bottle.
- Add 10-20 ml of vinegar and place the lid back on.
-
Hold the mouth of the small bottle over the large CO2 container and pour the CO2 collecting in the small container into the large container. Hold the small bottle horizontal so that the vinegar does not spill into the bigger bottle, only the heavier carbon dioxide gas pours into the large container.
- Add more vinegar when the effervescence stops. Repeat 2-3 times until the bottle is full. If a burning match at the mouth of the bottle goes out immediately, the bottle is full.
- Secure the thermometer and close the lid tightly.

- Measure and record the starting temperature of both bottles.
- Switch on the heat source and measure the temperature increase in both bottles. You need to decide for yourself what time increments are appropriate and record these in the table.

RESULTS:
Complete the following table.
|
Time (minutes) |
Temperature of air bottle (°C) |
Temperature of CO2 bottle (°C) |
Represent your results by drawing a graph for each of the experiments to show how the temperature for each bottle changed over time. You need to decide what values to use for each axis. Label the axes clearly and provide a heading for each graph.
Learners should draw both graphs on the same set of axes, or if not, then both graphs should have the same scales so that the graphs can be compared,
An example graph is shown here:
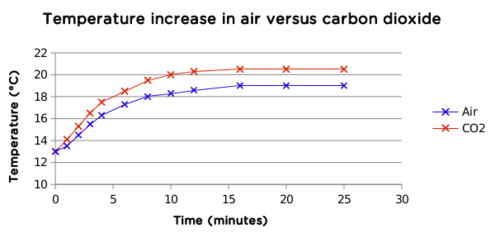
Both graphs follow a similar trend, but the carbon dioxide graph flattens out at a higher temperature. This means that carbon dioxide has trapped more heat than air. Although it is only a small difference (1,5°C), it is significant. The temperature change takes place quite quickly (within 20 minutes) and can easily be demonstrated within one lesson.
What have you observed?
The temperature in the carbon dioxide bottle increased more than in the bottle with air.
In both cases the temperature increased quickly, and then stabilised from about 15 minutes.
CONCLUSION:
What do you conclude for your experiment?
The carbon dioxide has trapped more heat than the air.
A demonstration of this experiment can be found here.
Extension investigation: What factors make the temperature of the atmosphere increase faster?
This is an optional extension in which learners must design their own experiment to answer one of the following questions. They can then write up an experimental report. You can use Assessment Rubric 2 to assess learners' work.
Design your own investigation to answer one or more of the following questions. Use the experiment above to guide your experimental set-up.
- Does dark soil make the temperature increase faster?
- Does water vapour make the temperature increase faster?
- Does the thickness of the layer of gases make the temperature increase faster?
- Does the presence of dust/aerosols make the temperature increase faster?
- Does the distance of the Sun make the temperature increase faster?
Global warming
What do you think will happen if the levels of carbon dioxide and other greenhouse gases increase? Think about what you discovered in the last investigation and look at the diagram of the greenhouse effect again. Write your answer below.
Learners should have seen from the last investigation that if the levels of carbon dioxide and other greenhouse gases increase in the atmosphere, then the temperature will increase as greenhouse gases trap more heat.
Climate change.
Mass extinctions.
If there are more greenhouse gases in the atmosphere, more ultraviolet radiation will be trapped and the Earth will heat up. This will result in more of the polar ice melting than usual. Even a one degree difference in the average temperature has an effect on the melting of polar ice. If more ice than usual melts, the water levels in the oceans will rise and low-lying areas could flood.
A change in the temperature will also result in a change in weather patterns. More rain will fall in some areas, and less in others. If this change is permanent, it is called climate change, and if it occurs on a worldwide scale it is called global climate change, which is what is being discussed here.
Global warming affects weather patterns which in turn has a knock-on effect on agriculture and food production. This has an impact on food production and can lead to food shortage for humans and animals. Long term climate change can lead to the extinction of plants and animals, which are unable to adapt to changed conditions.
The levels of greenhouse gases vary naturally over time. A question that scientists often ask is whether the concentration of greenhouse gases is rising more than it would naturally as a result of human activities? How do you think this can be investigated?
Since the industrial revolution humans have burned more fossil fuels than ever before. Human activities have resulted in the increase of carbon dioxide emissions over time. Carbon dioxide is therefore the main greenhouse gas under discussion amongst scientists and environmentalist. The following investigation will look at the levels of carbon dioxide over thousands of years.
Global warming 101.
Ice core analysis
Carbon dioxide is trapped in the ice which forms at the poles. As the ice is compacted and becomes thicker over thousands of years, the carbon dioxide remains trapped. The levels of carbon dioxide in ice can be determined by analysing the ice cores. A research team in Antarctica drilled an ice core containing ice from 160 000 years ago. They analysed the ice for carbon dioxide and presented their data in the following table.
Climate science: Antarctica's ice cores (video).
An ice core is a core sample from the accumulation of snow and ice over many years that have recrystallized and have trapped air bubbles from previous time periods.


Results from the ice core analysis.
ppm stands for 'parts per million'.
|
Number of years ago |
CO 2 levels (ppm ) |
|
160 000 |
190 |
|
150 000 |
205 |
|
140 000 |
240 |
|
130 000 |
280 |
|
120 000 |
278 |
|
110 000 |
240 |
|
100 000 |
225 |
|
90 000 |
230 |
|
80 000 |
220 |
|
70 000 |
250 |
|
60 000 |
190 |
|
50 000 |
220 |
|
40 000 |
180 |
|
30 000 |
225 |
|
20 000 |
200 |
|
10 000 |
260 |
|
8160 |
280 |
|
0 |
387 |
INVESTIGATIVE QUESTION: Write down an investigative question for this study.
How has the level of carbon dioxide in the atmosphere changed over the past 160 000 years?
ANALYSIS:
Draw an accurate graph to represent your data. You need to choose your own set of axes, and label them appropriately.
An example of the graph that learners produce is shown here:
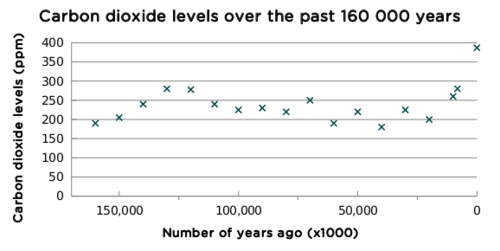
What is the link between the levels of CO2, core ice and global warming?
Pockets of CO2 are trapped in the ice. These pockets provide a record of CO2 levels in the air atmosphere at that time. When we analyse ice from 160 000 ago, we can determine the level of CO2 present in the atmosphere at that time. Higher levels of CO2 means that the greenhouse effect is stronger and the Earth heats up more.
CONCLUSION:
Write down a conclusion for this investigation.
The levels of CO2 have risen/doubled over the past 160 000 years.
What is the impact of global warming on the planet?
Increase in ocean temperatures, ice caps melting, increase in sea level, change in weather patterns, change in crop production, possible food scarcity/hunger, some areas become wetter, others dryer, animal and plant species may go extinct.
Could global warming, a problem here on Earth, be the solution to making Mars a habitable planet?
- The layer of gases around the Earth is called the atmosphere.
- The density of the gas molecules decreases as the distance from the Earth increases - the further away from the Earth you travel, the fewer gas molecules there are.
- The atmosphere can be divided into different layers - the troposphere, stratosphere, mesosphere and thermosphere.
- The exosphere is the uppermost layer directly above the thermosphere, where the gases thin out and the atmosphere merges with space. It is considered part of outer space.
- The troposphere is the densest layer, has the highest air pressure and is closest to the surface of the Earth. It is on average about 12 km thick and temperature decreases with altitude.
-
The stratosphere stretches from 12 - 50 km and contains the ozone layer. Aeroplanes fly in this layer because the air is more stable. Temperature increases with altitude, from -60°C to 0°C.
-
The mesosphere stretches between 50 - 80 km. The air is very thin. Meteorites usually burn up in the mesosphere. Temperature decreases with altitude from 0°C to -90°C.
-
The thermosphere stretches up to 480 - 600 km. It absorbs ultraviolet light and X-rays. Temperature increases with altitude and can reach 1500°C.
- The ionosphere is the layer where molecules are ionised by the Sun's ultraviolet light. Radio waves can be transmitted and reflected due to the ionised layer.
- The greenhouse effect is a natural phenomenon - it warms the atmosphere sufficiently to sustain life.
- Greenhouse gases trap the re-radiation from Earth's surface and reflect it back to the Earth (like inside a greenhouse).
- The most common greenhouse gases are carbon dioxide, water vapour and methane.
- An increase in greenhouse gases leads to global warming.
- Global warming is an increase in the average temperature of the atmosphere.
- Global warming is a potentially life threatening situation on Earth. It can lead to climate change, rising sea levels, food shortages and the extinction of organisms on Earth.
Concept map
Through the past 2-3 years you have come across concept maps in Natural Sciences. Use what you know about concept maps and design a map for this chapter. You must add terms and examples to the list. Remember to use linking words between concepts, and arrows to indicate the direction in which information is read. Plan your concept map on rough paper first before drawing the final one into your workbook. Use the following terms to help you with your map:
- atmosphere
- layers
- mesosphere
- thermosphere
- troposphere
- stratosphere
- weather
- ozone
- satellites
- radio waves
- global warming
- greenhouse gases
- greenhouse effect
- oxygen
- carbon dioxide
- water vapour

It is important that learners complete this concept map to consolidate what they have learned. They can either do it as a homework exercise or if enough time is available, a whole period can be dedicated to this activity. Learners have been exposed to many concept maps up until now and have had to make their own from scratch for some of the smaller chapters. Learners now have to put these skills into practice for a more complex concept map. This will be good preparation for learners' examinations. There are no right and wrong answers for the concept map. An example concept map is provided here. If learners are battling, you could use this following map as a guide and with the class, draw a concept map on the board. However, do not just draw it up for learners to copy down. Learners must be actively involved in the construction. You may also come up with alternative ways of presenting the concepts.
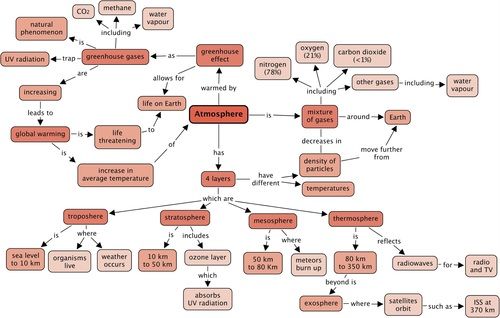
Revision questions
The following graph shows the variation in temperature as you move further away from the Earth. Study it and answer the questions that follow.
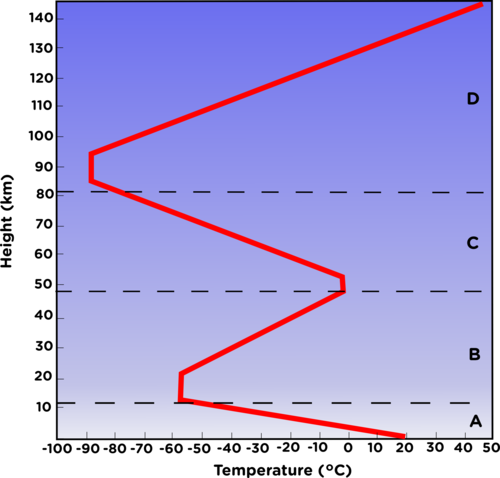
-
Give labels for A-D, the layers of the atmosphere. [4 marks]
-
Describe the temperature change in each of the layers. [4 marks]
-
Explain why the temperature changes as you move further away from the Earth in Layer A? [2 marks]
-
In which layer is the density of gas the highest? Give a reason for your answer. [2 marks]
-
In which layer(s) can life survive? Give two reasons for your answer. [3 marks]
-
In which layer are satellites found? Write only A, B, C or D. [1 mark]
-
In which layer are meteors found? Write only A, B, C or D. [1 mark]
-
In which layer radio waves reflected? Write only A, B, C or D. [1 mark]
-
In which layer is weather observed? Write only A, B, C or D. [1 mark]
-
In which layer is the aurora found? Write only A, B, C or D. [1 mark]
-
In which layer do jet aeroplanes travel? Write only A, B, C or D. [1 mark]
-
In which layer are lightning and storms found? Write only A, B, C or D. [1 mark]
-
In which layer is ozone found? Write only A, B, C or D. [1 mark]
-
A - troposphere
B - stratosphere
C - mesosphere
D - thermosphere
-
A - The temperature decreases with altitude from about 16°C to -60°C.
B - The temperature increases with altitude from -60°C to 0°C.
C - The temperature decreases with altitude from 0°C to -90°C.
D - The temperature increases with altitude from -90°C to 1000°C.
-
The troposphere is heated mainly through radiation of the Sun's energy by the Earth. The further you move away from the Earth, the less radiation energy there is to heat the atmosphere. Temperature will therefore decrease with altitude.
-
The troposphere because it is the closest to the Earth and the Earth's gravity pulls the gases in the atmosphere towards the surface.
-
The troposphere. It is warm enough and has enough oxygen for respiration. At this level, life is also protected from UV radiation.
-
D
-
C
-
D
-
A
-
D
-
B
-
A
-
B
Venus and Mars contains equal amounts of carbon dioxide, yet the temperature on the surfaces of these two planets are very different. Explain why. [4 marks]
Venus is closer to the Sun. Both have CO2, but Venus' atmosphere is much denser. The greenhouse effect is much more dominant, resulting in a much hotter surface temperature. Mars has lots of CO2, but it is not very dense and therefore the greenhouse effect is not observed.
Earth is the only planet that we know of that sustains life. What makes Earth's atmosphere suitable to sustain life? [2 marks]
Earth has enough oxygen and not a lot of CO2. There are just enough greenhouse gases to ensure that the Earth is kept warm enough. It has just the right balance between CO2 and oxygen/other gases.
Scientific evidence seems to point to the fact that carbon dioxide levels have increased steadily over the past 200 years.
-
Why would the levels of carbon dioxide have been increasing over the past 200 years? [2 marks]
-
What is global warming? [1 mark]
-
What are the long term effects of an increase in carbon dioxide on life on Earth? [4 marks]
-
Industries, human activities, burning of fossil fuels, more cars/vehicles (any two).
-
It is the gradual increase in the temperature of the Earth's atmosphere.
-
Higher levels of CO2 means that the greenhouse effect will increase and temperatures on Earth will rise. If this continues over a long time, ice will melt, water levels will rise, coastlines will change, weather patterns will be influenced, crop production will be affected, and it could lead to the extinction of animal and plant species that are unable to adapt.
Total [36 marks]
