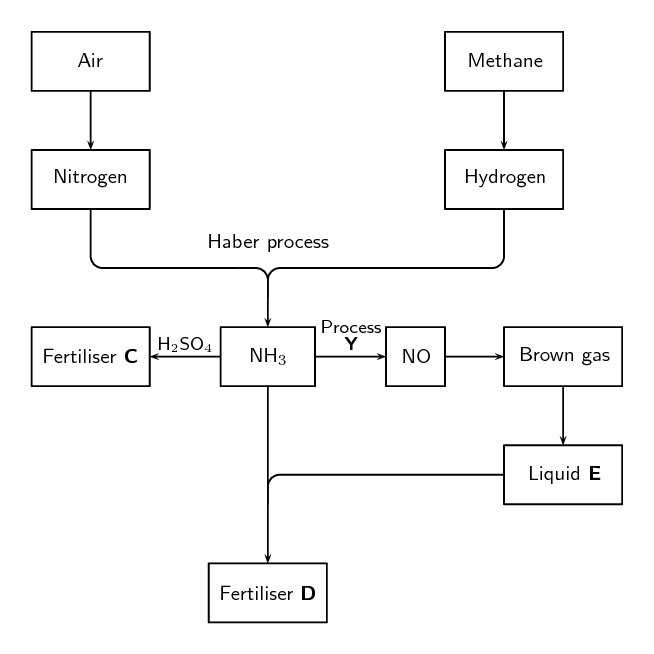
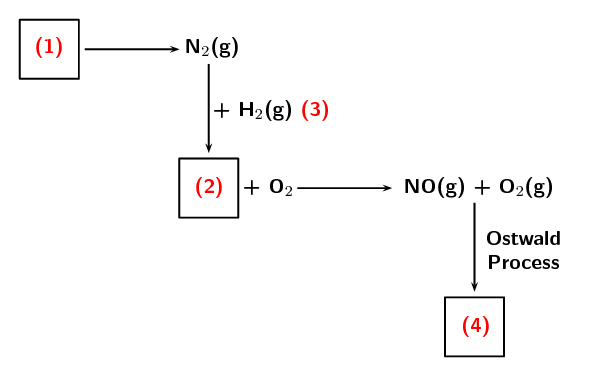
The following extract comes from an article on fertilisers:
A world without food for its people and a world with an environment poisoned through the actions of man are two contributing factors towards a disaster scenario.
Do you agree with this statement? Write down at least three arguments to substantiate your answer.
Learners give their own opinion.